 In the normal state of an atom, the number of electrons is equal to the number of protons. No orbit cannot accommodate more than 18 electrons. 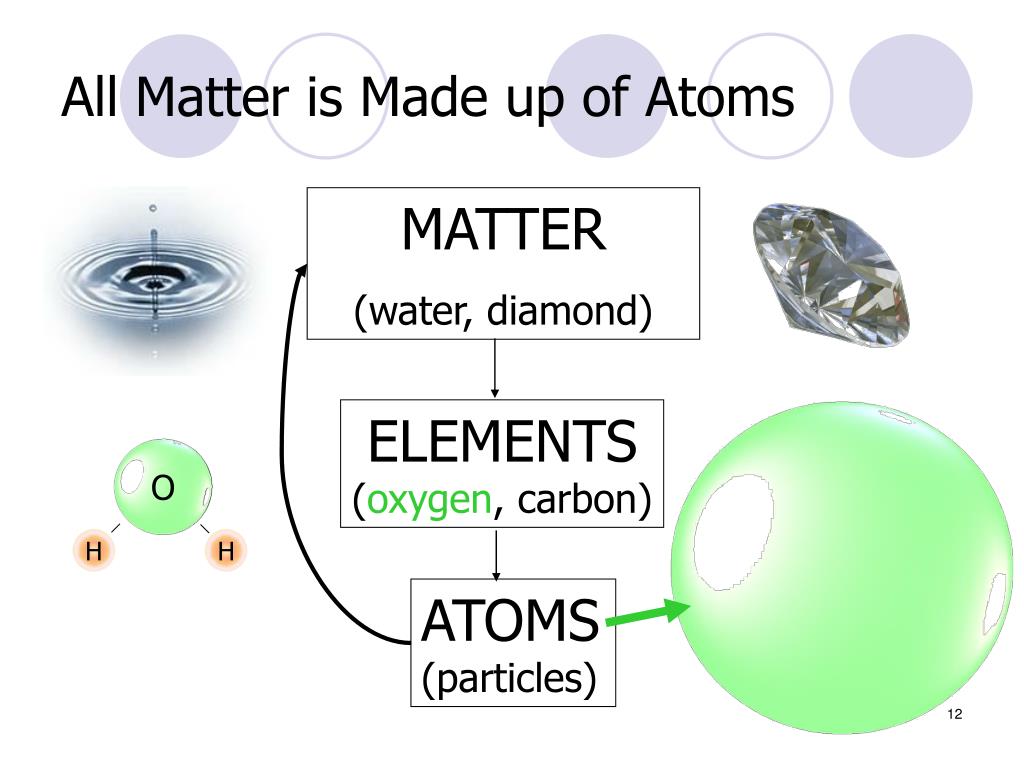
The outermost (last) orbit can have maximum 8 electrons. Thus, the first orbit has 2 electrons, the second orbit has 8 electrons and so on. The magnitude of charge on a proton is equal to $\mathrm$, where n is the number of the orbit. The proton is a positively charged particle. The nucleus is the central part of an atom and it contains two subatomic particles namely protons and neutrons. Basically, atoms are the sub-parts of molecules. The atom is the basic building block of the substance. For example, water is a compound because its molecules contains two atoms of hydrogen and one atom of oxygen. The substance whose molecules consist of different kind of atoms is called compound. For example, oxygen is an element because its molecule has two atoms of same type. The substance whose molecules consist of same type of atoms is called element. Where, a molecule is in turn made up of minute particles called atoms.Īccording to the electron theory of matter, This theory states that all substances whether solid, liquid or gas is composed of small particles called molecules. The nature of electricity can be easily explained by the electron theory of matter. This article is meant for explaining the concept of the electron theory of matter and the atom. 
The electron theory of matter is the result of experiments and researches conducted by many scientists like J. The only theory that has explained it successfully is the electron theory of matter. Although the study of electricity has been attracting the attention of scientists for several hundred years, and there were several experiments and theories developed to understand the nature of electricity. The electron theory of matter is one of the most successful and experimentally proved theory that can explain the nature of electricity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
